|
Which of the following questions about the compound can be answered using the results of the analysis?ī) What is the molar mass of the compound?Ĭ) What is the chemical stability of the compound?ĭ) What is the empirical formula of the compound? The results showed that the sample contained 36.0g of C and 6.0g of H. We can solve the question 'What is the empirical formula of the compound?' The answer is CH2Ī 42.0g sample of compound containing only C and H was analyzed. The molar mass of CO2 is 12 + 2(16) = 44g/mol Since we have the number of moles of CO2 now, we need to know the molar mass to enable us get the mass Since 6 moles of CO2 were produced from one mole of glucose, the number of moles of glucose produced is thus 6 * 0.25 = 1.5 moles Now we proceed to get the number of moles of CO2 produced. The number of moles is thus 45/180 = 0.25 mole 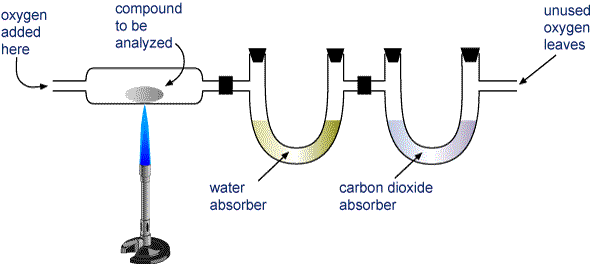
This can be obtained by dividing the mass by the molar mass. 
Firstly, we get the number of moles of glucose reacted. Now, we need to get what happened actually. Here we have glucose reacting with oxygen to give carbon iv oxide and water plus energy.įrom the chemical reaction, we can see that 1 mole of glucose yielded 6 moles of carbon iv oxide. The first step to solving this problem is by writing a balanced chemical reaction. 
44 g of CO2 - 12 g of carbonġ8 g of water - 1 g of hydrogen
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
